Time-dependent measurements - Homepage WEE-Solve GmbH
Main menu:
- Home
- Menütrennlinie 7
- About us
- Menütrennlinie 1
- Fractionation
- Menütrennlinie 4
- Rheology
- Menütrennlinie 5
- Contract research
- Menütrennlinie 6
- Analysis
- Menütrennlinie 8
- Contact
Time-dependent measurements
Time-
For a simple reaction of the components A, B, and C to the product D, i.e. without any intermediate step or side-
The result of it is the velocity law of the temporal decreasing of component A. Thereby k is the reaction velocity constant, which depends on the ambient parameter (e.g. temperature, pressure, catalyst influence etc.).
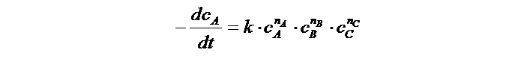
The sum of the superscripts n is called the reaction order. For the determination of the actual reaction progress, the differential equation has to be solved. By plotting the time flow of the concentration, we can see that for identical velocity constants k the gradient become shallower.
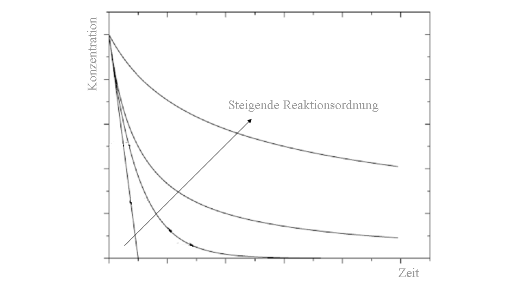
Temporal decrease of the concentration for different reaction orders with identical velocity constants k
According as the absorbing substance is reacting agent or a product of the investigated reaction, we can see at a time-
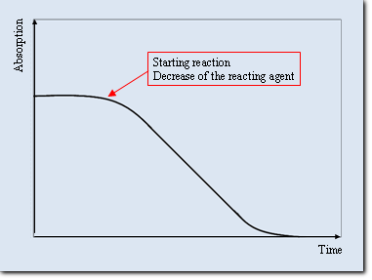
Time-
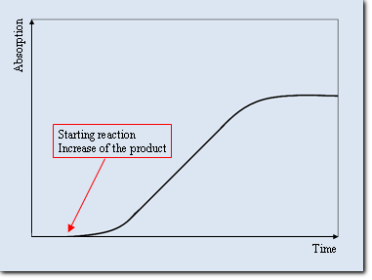
Time-
